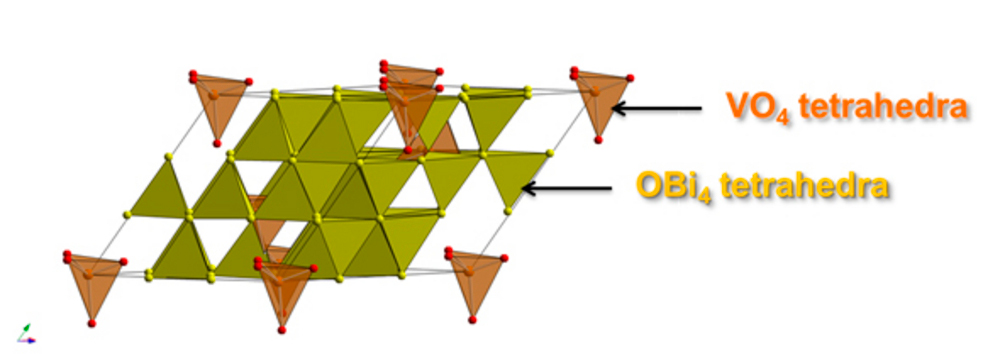
Given the ever-increasing need for cleaner, more efficient energy sources, materials scientists are constantly searching for new ways to understand and exploit the properties of functional materials to create more environmentally-friendly electrical devices. A team of scientists from the Institut Laue-Langevin (ILL) and Durham University, UK, have used neutron scattering techniques to characterise the material properties of oxide ion conductors, solids with highly mobile oxide ions, which can be used for several energy and environmental applications, such as oxygen sensors and pumps or solid oxide fuel cells (SOFCs).
The study, which was recently published in the Journal of the American Chemical Society, is part of an on-going collaboration between Professor Ivana Evans at Durham University, and Dr Andrea Piovano and Professor Mark Johnson at the ILL.
“Our ultimate goal is to understand the properties and exploitable behaviour of oxide ion conductors at the atomic level,” says Professor Ivana Evans. “This means understanding their structure and dynamics, and how they evolve with temperature. By examining the relationship between the atomic-level and macroscopic properties, we can apply this knowledge to develop new and improved materials.”
One of the most promising applications of oxide ion conductors is as electrolytes in solid oxide fuel cells (SOFCs), devices for efficient electricity generation. Oxide ion conductors have several properties which make them suitable for use in fuel cells. The solid electrolytes in SOFCs allow the movement of ions through the device without the need for a liquid or soft membrane separating the electrodes as in traditional fuel cells – this results in higher current densities and reduced risk of current leakage. SOFCs allow efficient energy conversion from chemical energy to electricity without creating environmentally-noxious by-products. They can also work with a variety of fuels and are less affected by possible impurities present in them. “To be able to harness and optimise these properties, we need to understand how the ion conduction mechanisms operate at the microscopic level,” says Dr Andrea Piovano.

About ILL
The Institut Laue-Langevin (ILL) is an international research organisation that is a world leader in Neutron Science. Since its foundation in 1967, the Institute is a shining example of scientific cooperation. Presently 10 European countries (Spain, Switzerland, Austria, Italy, Czech Republic, Sweden, Belgium, Poland and Slovakia) ensure the necessary financial support for the ILL operation under the governance of 3 Associate Member countries: France, Germany and The United Kingdom. The research conducted at the ILL is dedicated to fundamental research (60%) as well as societal challenges research (40%). It covers a wide range of disciplines such as biology, (green) chemistry, materials science, condensed matter physics, as well as nuclear and particle physics.
© ILL Given the ever-increasing need for cleaner, more efficient energy sources, materials scientists are constantly searching for new ways to understand and exploit the properties of functional materials to create more environmentally-friendly electrical devices. A team of scientists from the Institut Laue-Langevin (ILL) and Durham University, UK, have used neutron scattering techniques to characterise […]